Absolute Entropy at 298 K (J/ (K mol)) NO (g) 211 O2 (g) 205 NO2 (9) 240 (a) Using the information in the table above, calculate the value of AS for the reaction. 
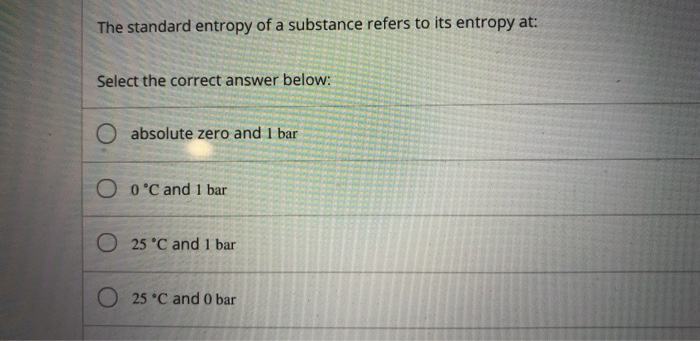
Finally, substances with strong hydrogen bonds have lower values of S°, which reflects a more ordered structure. The third law of thermodynamics defines absolute zero on the entropy scale. 2 NO (g) + O2 (g) 2 NO2 (g) AH 112 kJ Answer the following questions about the reaction represented above at 298 K. Entropy is a value you can get in a thermodynamics data if working at 25C. For example, compare the S° values for CH 3OH(l) and CH 3CH 2OH(l). What is the absolute entropy of O2 (g) C2H4(g) + 3O(g)->2CO2(g)+6H2O(g). Onnes helium liquefaction cascade: chloromethane, ethylene, oxygen, liquid. Similarly, the absolute entropy of a substance tends to increase with increasing molecular complexity because the number of available microstates increases with molecular complexity. All systems in thermodynamic equilibrium at absolute zero have vanishing entropy. As a system approaches absolute zero temperature, all processes cease and the entropy of the system approaches a universal constant (that can be taken to be zero). Soft crystalline substances and those with larger atoms tend to have higher entropies because of increased molecular motion and disorder. The third law of thermodynamics is an axiom of nature regarding entropy and the impossibility of reaching absolute zero of temperature (Nernst’s theorem, 1906). In contrast, graphite, the softer, less rigid allotrope of carbon, has a higher S° due to more disorder in the crystal. Among crystalline materials, those with the lowest entropies tend to be rigid crystals composed of small atoms linked by strong, highly directional bonds, such as diamond. \( \newcommand\) also reveals that substances with similar molecular structures tend to have similar S° values. Unlike enthalpy or internal energy, it is possible to obtain absolute entropy values by measuring the entropy change that occurs between the reference point.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
